Recently, a research paper titled “Gut microbiota-derived trimethylamine N-oxide contributes to cardiomyocyte pyroptosis and cardiac injury via the tRF-Glu-ANT1-GSDMD axis” was published online in the journal *Cellular & Molecular Biology Letters* by a team led by Professor Diao Hongyan and Assistant Researcher Wang Tao at the Jinan Microecological Biomedicine Shandong Laboratory. This study reveals the key role of the regulatory mechanism whereby gut microbiota metabolites activate the tRF-Glu-ANT1-GSDMD signalling axis in cellular inflammatory cell death, providing a new perspective for the development of gut microbiota-targeted therapeutic strategies. The Jinan Microecological Biomedicine Shandong Laboratory is listed as the first author institution for this paper.
Ischaemic heart disease is one of the leading causes of morbidity and mortality worldwide, primarily characterised by irreversible loss of cardiomyocyte function, which ultimately progresses to heart failure, severely impacting patients’ quality of life. Previous studies have shown that traditional cardiovascular risk factors do not underlie the pathogenesis of all cardiovascular diseases; rather, impaired intestinal barrier function leads to bacterial translocation and systemic inflammation, thereby exacerbating myocardial damage. Trimethylamine N-oxide (TMAO) is a gut metabolite derived from phosphatidylcholine; elevated plasma TMAO levels are independently associated with an increased risk of adverse cardiovascular outcomes, including atherosclerosis, heart failure and vascular ageing. However, the mechanisms underlying the interaction between the cardiac and gut microbiomes remain to be fully elucidated.
The research team first established a mouse model of myocardial infarction and found that a high-choline diet could increase plasma TMAO levels, thereby disrupting the intestinal barrier and exacerbating pyroptosis in cardiomyocytes and the progression of myocardial injury. tRNA-derived fragments (tRFs) and tRNA-like RNAs (tiRNAs), also known as tsRNAs, are a class of tRNA-derived regulatory RNAs formed by specific cleavage. They exert biological functions such as gene expression regulation, cell cycle regulation, chromatin regulation and epigenetic modification through various mechanisms. Using tRF & tiRNA-seq sequencing technology, the researchers found that under high-choline dietary conditions, the expression level of the tRF-1:31-Glu-TTC-2 (tRF-Glu) derived from tsRNAs was the most significant, emerging as a key intervention target for regulating cardiomyocyte pyroptosis. The absence of tRF-Glu significantly improved TMAO-induced myocardial fibrosis and the deterioration of cardiac function. Mechanistically, tRF-Glu directly binds to the mitochondrial inner membrane protein ANT1 (adenylate transposase 1) and stabilises its expression by inhibiting ubiquitination. Furthermore, inhibition of ANT1 in cardiomyocytes significantly blocked TMAO-induced cellular ROS production, restored membrane potential, and prevented mtDNA leakage.
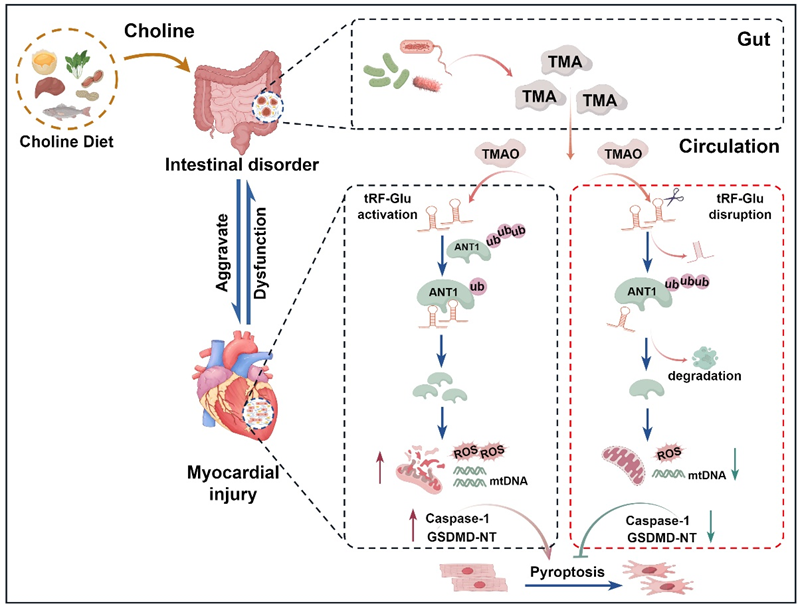
Fig. Schematic diagram of the mechanism by which the gut metabolite trimethylamine oxide regulates myocardial injury via tRF-Glu
Research Implications: The research team revealed that tRF-Glu participates in the regulation of pyroptosis in cardiomyocytes by inhibiting the ubiquitination of ANT1, thereby activating GSDMD expression and the release of mtDNA. This discovery establishes the TMAO/tRF-Glu/ANT1 signalling axis as a key intervention target in ‘gut-heart’ interactions, opening up new avenues for the prevention and treatment of myocardial injury.
Wang Tao, Assistant Researcher at the Jinan Microecological Biomedicine Shandong Laboratory, is the first author of this paper; Professor Diao Hongyan is the corresponding author; and the Jinan Laboratory is the lead institution. This research was supported by the National Key Research and Development Programme (2021YFA1301100, 2021YFA1301101), the Shandong Provincial Natural Science Foundation (ZR2024QH017, ZR2024QH318, ZR2024LZL003), the National Natural Science Foundation of China (82400348), the Shandong Provincial Laboratory Programme (SYS202202), and research projects from the Jinan Microecological Biomedicine Shandong Laboratory (JNL-2023009Q, JNL-2025002B, JNL-2025001A).