Recently, the team led by Lü Longxian, Director of the Pilot-Scale and Transformation Platform at Jinan Laboratory, published an online review titled ‘Next-Generation Microencapsulation Technologies for Probiotic Protection and Precision Delivery’ in Microbial Biotechnology. This review systematically outlines the health benefits of probiotics, the necessity of microencapsulation technology, and the environmental stress factors it addresses. It focuses on analysing how microencapsulation enhances probiotic tolerance to extreme environments, while thoroughly exploring its mechanisms for countering environmental stress and achieving controlled release. Concurrently, the paper briefly evaluates the safety of encapsulated probiotics and analyses key challenges in their industrialisation process. It concludes with a systematic outlook on future directions for microencapsulated probiotics in industrial and commercial applications.

The numerous health benefits of probiotics are well-established, primarily encompassing improved gastrointestinal function, regulation of microbial balance, and enhanced immune defence. However, during oral delivery, probiotics encounter multiple stresses including gastric acid, bile salts, and the localised pathological environment of the intestine, leading to a significant reduction in survival rates that limits the full expression of their therapeutic effects. To address these challenges, microencapsulation technology has emerged as an innovative and adaptable strategy for protecting and delivering probiotics.
Most encapsulation techniques, such as extrusion, layer-by-layer self-assembly, or self-assembly methods, involve encasing probiotics within a biocompatible matrix. Encapsulated probiotics provide protection against environmental and digestive stresses through mechanisms including physical shielding, pH-responsive polymer dissolution, redox-sensitive degradation, and enzyme-triggered breakdown, all while preserving bacterial viability. Furthermore, these techniques enhance probiotic adhesion within the gut and enable controlled, targeted release. This not only improves survival rates during production, storage, and transport but also holds broader application prospects in personalised nutrition and precision medicine. By designing functional materials tailored to specific disease characteristics, this approach enhances targeting to pathological sites, thereby further improving therapeutic efficacy.
Despite promising prospects, translating microencapsulated probiotics into commercial and clinical applications faces several obstacles. These include the absence of standardised safety and quality assessment protocols, technical challenges in maintaining capsule integrity during large-scale production, and the lack of globally harmonised regulatory guidelines. To advance this field, future efforts should prioritise developing novel, biocompatible, and cost-effective encapsulation materials, alongside scalable production processes that ensure high probiotic viability and functional stability.
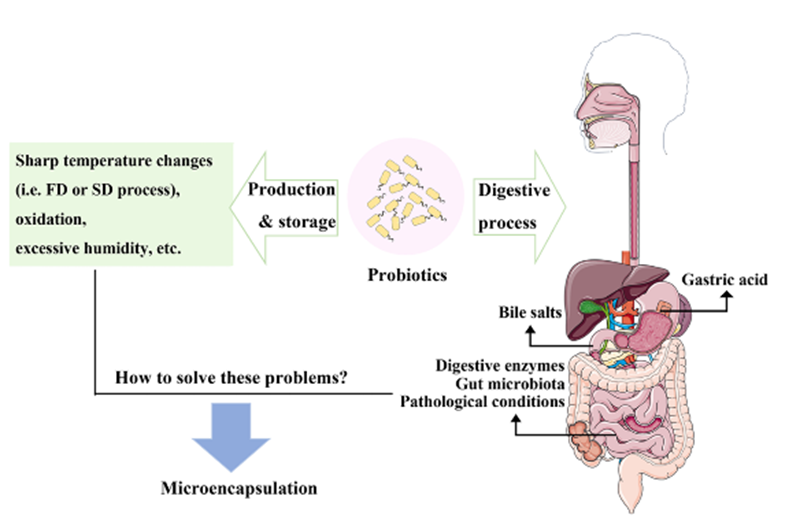
Microencapsulation serves as an effective strategy for enhancing probiotic gastrointestinal tolerance and targeted delivery.
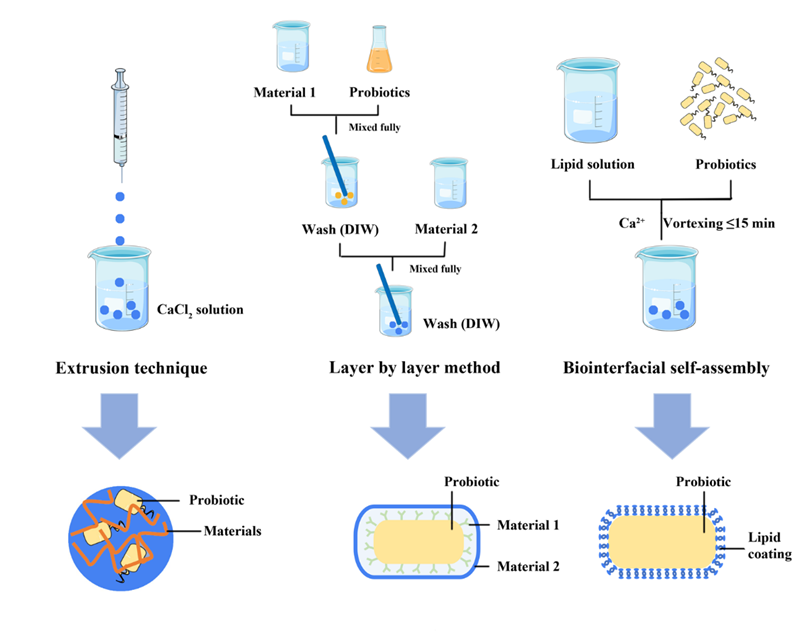
Primary methods of microencapsulation
Research Insights
Microencapsulation technology has emerged as a transformative strategy for enhancing probiotic survival rates by shielding them from external stressors. Leveraging advanced encapsulation materials and innovative processes, this technology significantly enhances probiotic tolerance to thermal, oxidative, and gastrointestinal stresses, enabling precise intestinal targeted release and efficient colonisation. By integrating cutting-edge research with practical applications, microencapsulation demonstrates immense potential in advancing probiotic therapies towards safer, more effective, and personalised treatments.
This review was supported by the National Key R&D Programme (2023YFC2506000, 2022YFC2304500), Shandong Provincial Laboratory Project (SYS202202), and Jinan Laboratory internal projects (JNL-2022009B, JNL-2022047D).
Original link: https://doi.org/10.1111/1751-7915.70305