Recently, Professor Wu Nanping's team at the Jinan Microecological Biomedicine Shandong Laboratory Sciences has achieved significant progress in phage therapy for multidrug-resistant Klebsiella pneumoniae infections. In vitro and in vivo evaluations in mice demonstrated the phage vB_KpnP-6K2 exhibits outstanding therapeutic efficacy against Klebsiella pneumoniae infections, providing crucial theoretical support for subsequent applications of phage therapy. The study, titled ‘A lytic bacteriophage vB_KpnP-6K2 inhibits ST11-KL64 Klebsiella pneumoniae induced cell death and inflammatory response’, has been published.

Klebsiella pneumoniae ranks among the primary pathogens causing opportunistic infections in hospitalised patients, with infections frequently exhibiting drug resistance. This resilience stems largely from the bacterium's capacity to acquire exogenous genetic elements encoding resistance and hypervirulence traits. As a major driver of the global antibiotic resistance crisis, K. pneumoniae causes over 100,000 deaths worldwide annually. Among these, strains resistant to carbapenems and third-generation cephalosporins pose an especially severe threat to public health.
The research team isolated a phage, vB_KpnP-6K2, from hospital wastewater that specifically targets the multidrug-resistant Klebsiella pneumoniae strain ST11-KL64. This phage exhibits favourable biological characteristics. Pneumonia and lung tissue damage caused by Klebsiella pneumoniae infection are major contributors to disease progression. This study identified monocytes as key cells in the inflammation induced by Klebsiella pneumoniae infection. Phage vB_KpnP-6K2 significantly reduced both the inflammatory response and cell death induced by Klebsiella pneumoniae infection.
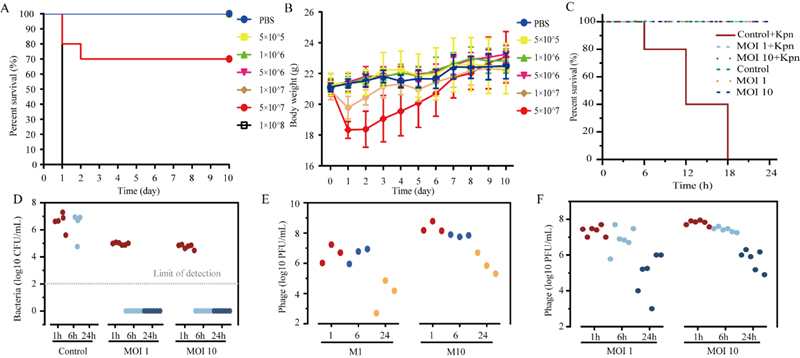
Furthermore, the research team established a mouse model of K. pneumoniae bloodstream infection to evaluate the therapeutic efficacy of phage vB_KpnP-6K2. Experimental results demonstrated that phage treatment effectively cleared K. pneumoniae from the bloodstream within 6 hours post-administration. As the bacteria were eliminated, the titre of phage vB_KpnP-6K2 in the mice's blood also decreased. This study further revealed that phages, as viruses capable of infecting bacteria, can also induce inflammatory responses and activate antiviral immunity.
Scientific Implications
The superior therapeutic efficacy demonstrated by phage vB_KpnP-6K2 in treating Klebsiella pneumoniae infections both in vitro and in mice not only indicates that phages could serve as an effective future strategy against ‘superbug’ infections but also provides crucial reference for the subsequent application and promotion of phage therapy. Future research should extend beyond examining phage-bacterial interactions to investigate therapeutic effects across diverse cell types within infected regions and at the individual level. This comprehensive approach will evaluate the interplay between phages, bacteria, and various cellular components, alongside their impact on disease progression and treatment outcomes.
This research was supported by the Shandong Provincial Natural Science Foundation (ZR2023QH546), the Jinan Microecological Biomedicine Shandong Laboratory Science (JNL-2022004Q, JNL-2023014D, JNL-2025005B), the National Key R&D Programme (2023YFC2506004), and the Shandong Provincial Laboratory Programme (SYS202202).
The first author of this paper is Dr Pan Zhaoyi from the Innovation Team for Microecology and Disease Research.
Pan Zhaoyi, Assistant Researcher, obtained his PhD in Immunopharmacology from the School of Pharmacy, Shandong University. His research focuses on viral infection and microbiome studies. In recent years, as principal investigator, he has undertaken research projects including the Jinan Laboratory Doctoral Start-up Fund, Laboratory Youth Fund, and Shandong Provincial Natural Science Foundation Youth Project. He has published over ten SCI papers as first author in journals such as PNAS and Virulence, and holds one invention patent and one utility model patent.