Recently, the team of Academician Gu Xiaosong and Professor Jiang Chunping from the Jinan Laboratory, in collaboration with Professor Ding Qiurong's team from the Shanghai Institute of Nutrition and Health, Chinese Academy of Sciences, has made significant progress in the research of metabolic dysfunction-associated steatohepatitis (MASH). For the first time, they revealed that selenoprotein H (SELENOH) acts as a coactivator of PPARα, linking selenium homeostasis to hepatic lipid metabolism and protecting against steatohepatitis. The study, titled "Selenoprotein H Functions as a PPARα Coactivator to Link Selenium Homeostasis to Hepatic Lipid Metabolism and Protect against Steatohepatitis," was published in Advanced Science (IF: 15.6).

Selenium, as an essential trace element, primarily exerts core biological functions such as redox regulation through 25 human selenoproteins. Its deficiency is closely associated with the progression of chronic liver diseases. MASH, a globally prevalent liver disease, currently has limited therapeutic options. Although studies have shown that selenium status significantly affects hepatic lipid metabolism—selenium deficiency leads to lipid metabolism disorders, while selenium supplementation effectively alleviates hepatic fat accumulation in animal models—the specific functions and mechanisms of most selenoproteins remain unclear.
Mechanistic Link Between Selenium Homeostasis and MASH
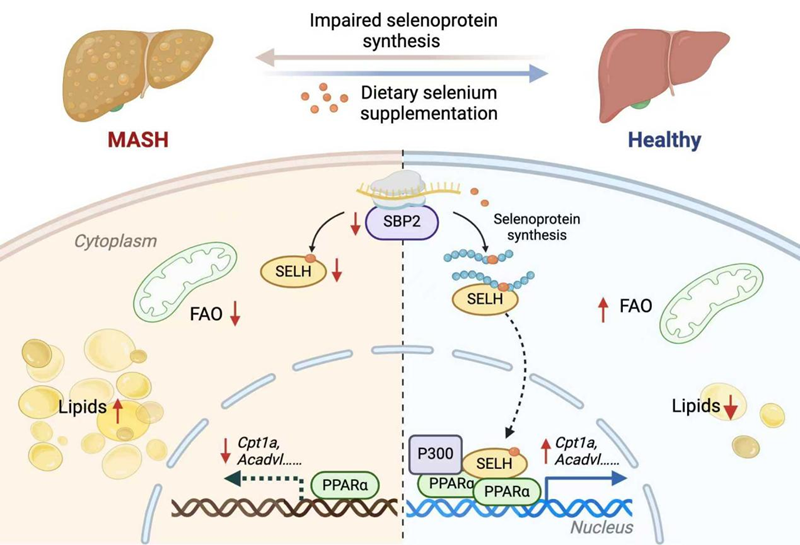
The study found that patients and animal models with MASH exhibit hepatic selenium imbalance, characterized by reduced selenium levels in serum and liver tissue and widespread impairment of selenoprotein synthesis. The key reason for this defect is the decreased expression of SECIS-binding protein 2 (SBP2), a critical regulator of selenoprotein biosynthesis. Supplementation with selenium or overexpression of SBP2 restored selenoprotein synthesis and effectively alleviated the pathological progression of MASH.
SELENOH Regulates Lipid Metabolism via PPARα
For the first time, the study elucidated that SELENOH is a key effector protein through which selenium exerts its liver-protective effects. In MASH, SELENOH expression is downregulated. Mechanistically, SELENOH does not function through traditional oxidoreductase activity but rather acts as a transcriptional coactivator. It binds to ligand-activated PPARα, promoting the recruitment of the coactivator P300, thereby driving the expression of genes involved in fatty acid oxidation and improving hepatic lipid metabolism. The SELENOH-PPARα axis represents a core pathway through which selenium supplementation ameliorates MASH.
Research Implications:
This study reveals for the first time that selenium deficiency exacerbates MASH progression through SBP2-mediated impairment of selenoprotein synthesis. It also innovatively uncovers the molecular mechanism by which SELENOH acts as a transcriptional coactivator of PPARα to regulate fatty acid oxidation in a non-redox manner, providing a new paradigm for understanding the interaction between trace element metabolism and nuclear receptor signaling. Future research should further elucidate the cell-specific functions of different selenoproteins in the liver microenvironment and explore safe and effective intervention strategies targeting this axis.
This research was supported by the Shandong Provincial Key Research and Development Program (Competitive Innovation Platform) (Grant No. 2025CXPT176), the Scientific Research Project of Jinan Microecological Biomedicine Shandong Laboratory (Grant No. JNL2025008B), and the Shandong Provincial Taishan Scholar Young Expert Program (Grant Nos. tsqn202507393, 202408310).