Recently, Professor Wu Nanping and Professor Zheng Shufa's team at the Jinan Microecological Biomedicine Shandong Laboratory Sciences focused on the pathogenesis and therapeutic challenges of inflammatory bowel disease (IBD). They systematically analysed the biological characteristics of the gut commensal bacterium Akkermansia muciniphila, elucidating its core mechanism for improving IBD by regulating intestinal immune and metabolic networks. The study further explored the application potential and challenges of this bacterium as a next-generation probiotic. The corresponding review, titled ‘The role of Akkermansia muciniphila in the regulation of inflammatory bowel disease: intestinal immunity and metabolism’, has been published in Frontiers in Immunology.

Inflammatory bowel disease (IBD), encompassing Crohn's disease (CD) and ulcerative colitis (UC), constitutes a chronic inflammatory condition of the gut. Its pathogenesis is closely associated with host immune dysregulation, gut microbiota imbalance, and compromised intestinal barrier function. In recent years, gut microbiota regulation has emerged as a research focus in IBD therapy. Among these, Akkermansia muciniphila, as a dominant microorganism in the intestinal mucus layer, possesses mucus-degrading capabilities and potential probiotic properties, positioning it as a key candidate for next-generation probiotics. Research indicates that the abundance of Akkermansia muciniphila is significantly altered in the intestines of IBD patients. Furthermore, both the live bacteria and its components (including outer membrane proteins and extracellular vesicles) demonstrate protective effects against IBD. This review systematically analyses its mechanisms of action in IBD from two core dimensions:
1. Intestinal Immune Regulatory Network
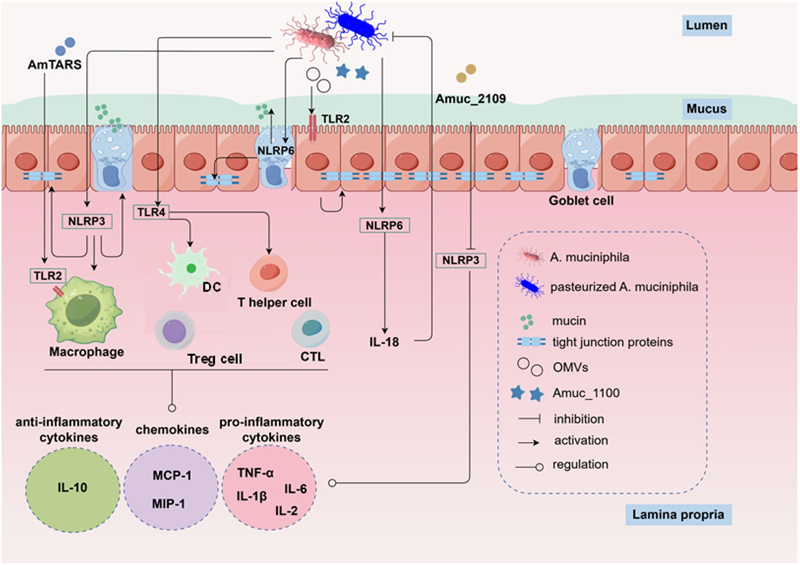
Akkermansia muciniphila reshapes intestinal immune balance through bidirectional interactions with immune cells, intestinal epithelial cells (IECs), and intestinal stem cells (ISCs):
· Components such as the outer membrane protein Amuc_1100, secretory threonyl-tRNA synthetase (AmTARS), and extracellular vesicles (OMVs)
(1) activate TLR2 and TLR4 pathways, regulating NLRP3 and NLRP6 inflammasome activity, thereby modulating immune cells to promote anti-inflammatory factor secretion, suppress pro-inflammatory factor release, and alleviate intestinal inflammation;
(2) it promotes tight junction protein assembly in the intestinal epithelium, stimulating goblet cell mucus secretion to repair mucus barrier damage;
(3) By activating signalling pathways such as Wnt/β-catenin, they promote intestinal stem cell (ISC) proliferation and intestinal epithelial regeneration, thereby enhancing the gut mucosal repair capacity.
2. Gut Metabolic Regulation Pathways
Akkermansia muciniphila participates in regulating three core metabolic pathways—tryptophan (Trp), short-chain fatty acids (SCFAs), and bile acids (BAs)—to synergistically improve IBD:
· (1) Tryptophan metabolism: Modulates immune responses and intestinal barrier function by inhibiting the kynurenine pathway and activating the aryl hydrocarbon receptor (AhR) pathway.
· (2) SCFA metabolism: Degrades mucin to produce SCFAs such as acetate and propionate, which activate GPR43 receptors to promote Treg cell differentiation and suppress intestinal inflammation;
· (3) Bile acid metabolism: By regulating the microbiota-bile acid axis, it influences secondary bile acid production, activates receptors such as FXR and TGR5, maintains intestinal barrier integrity, and modulates immune responses.
Research Implications:
This review systematically elucidates the dual immunometabolic mechanisms by which Akkermansia muciniphila modulates IBD, offering novel insights into gut microbiota-host interactions. Possessing multi-targeted, synergistic anti-inflammatory properties, Akkermansia muciniphila and its components demonstrate significant therapeutic potential for IBD. Nevertheless, its effects remain controversial, potentially exacerbating inflammation under specific conditions. Issues such as strain specificity, colonisation patterns, and clinical translation safety require urgent resolution. Future research should delve deeper into the differential actions and molecular mechanisms of various forms of Akkermansia muciniphila, providing theoretical underpinnings for developing precision-targeted microbial therapies.
This research received funding from the Jinan Microecological Biomedicine Shandong Laboratory (JNL-2023012Q, JNL-2022005Q), the Shandong Natural Science Foundation (ZR2023QB299), and the Taishan Scholar Award Programme (tsqnz20230627).