Recently, Professor Wu Nanping's team published a research paper entitled ‘Interrelationship between altered metabolites and the gut microbiota in people living with HIV with different immune responses to antiretroviral therapy’. The paper reveals the interrelationship between altered metabolites and the gut microbiota in people living with HIV with different immune responses to antiretroviral therapy.

Antiretroviral therapy effectively suppressed plasma HIV RNA levels to undetectable levels in HIV-infected patients, thereby promoting immune system recovery. This critical intervention significantly reduces the risk of opportunistic infections and improves the survival of HIV-infected individuals.
However, some infected individuals who fail to normalise their CD4+ T-cell counts even after sustained viral suppression, known as immune non-responders, are at higher risk of rapid disease progression and increased mortality compared to immune responders.
Recent studies have shown that HIV infection is associated with alterations in intestinal flora. At this stage, research has mainly focused on characterising the effects of HIV infection on host-microbe interactions in the intestinal tract, with relatively little research on the interactions between intestinal flora and its metabolites and the host, and relatively limited research on the interrelationships between immune markers, intestinal metabolites and intestinal flora.
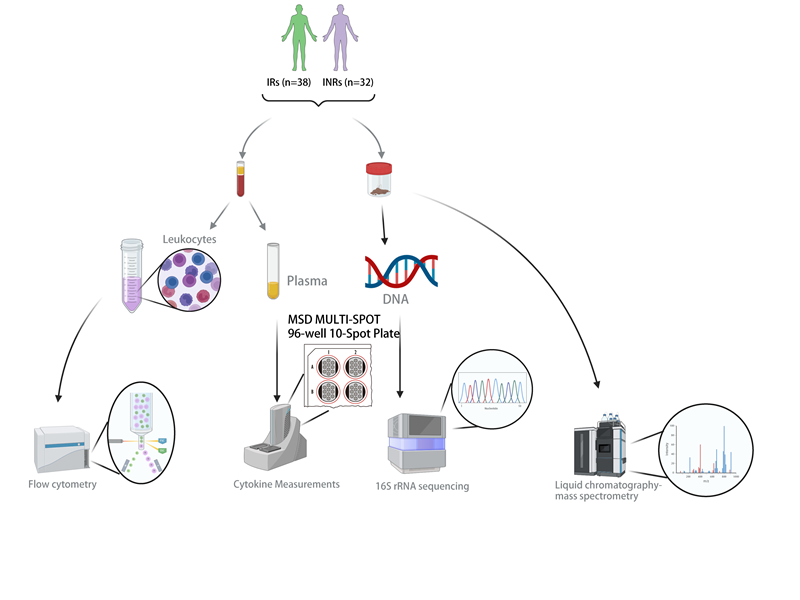
Based on the above research background, the research team relied on the previously established cohort of HIV-infected patients, using genomics, metabolomics and other technical means, and found that the composition of the gut flora of the immune-naïve patients was altered, and the diversity was reduced; the metabolic function of the gut flora showed a downward trend, in which the microbial function of the folate biosynthesis and biotin metabolism was significantly reduced; and the differences in the genus of the bacteria and the metabolites could distinguish between different immune statuses of the patients (AUC = 0.8). status (AUC = 0.8125).
Cellular experiments revealed that indole-3-acetyl-β-1-D-glucoside (AUC = 0.8931) significantly reduced the proportion of CD4+ CD57+, initial CD4+, CD8+CD57+, effector CD8+, and initial CD8+ T cells (P < 0.05), and the expression of inflammatory factor mRNAs such as IL-6, IL-17A, and IFN-γ (P < 0.001). P < 0.001).
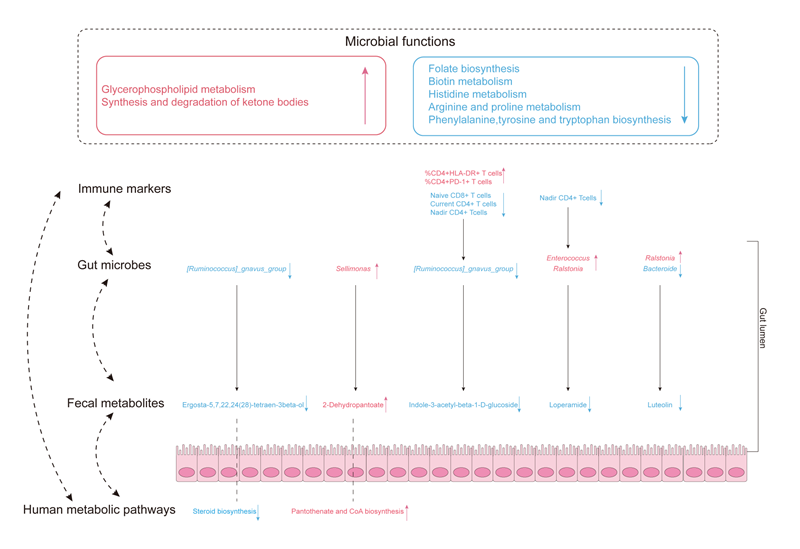
This study explored the interactions between gut flora and its metabolites with the host through genomics and metabolomics, and found that the immune unresponsive state not only disrupts the gut flora, but also the reduced metabolites play potential roles in immune activation and inflammatory responses, which will advance our understanding of immune recovery in immunodeficient patients from different perspectives, and explore new preventive or interventional measures.
Jinan Microecological Biomedicine Shandong Laboratory is the first completion unit of this paper. Xuebin Tian, a PhD student, is the first author, and Prof Nanping Wu is the corresponding author. This project was supported by the Jinan Provincial Laboratory of Microecology and Biomedicine (JNL-2022005B), Shandong Provincial Laboratory Project (SYS202202), and the National Key Research and Development Programme (2023YFC2506000, 2023YFC2506004).