On 14 March 2025, the team of Academician Xiaosong Gu, Deputy Director of Jinan Laboratory, Professor Chunping Jiang and PI Junhua Wu presented their research results on ‘Viral expression of NE/PPE enhances anti-colorectal cancer efficacy of oncolytic adenovirus by promoting TAM M1 polarisation to reverse insufficient adenovirus by promoting TAM M1 polarization to reverse insufficient effector memory/effector CD8+ T cell infiltration’ in Journal of Experimental & Clinical Cancer Research (IF: 11.4, CAS Region I). The team reported that adenoviral expression of NE/PPE enhances the therapeutic efficacy of lysogenic adenovirus against colorectal cancer by promoting the M1 polarisation of the tumour microenvironment and reversing the lack of effector memory/effector CD8+ T cell infiltration.

According to the International Agency for Research on Cancer (IARC) 2022 global statistics, colorectal cancer has the second highest mortality rate, constituting a significant health burden and global public health challenge. Clinical treatment of colorectal cancer usually involves a multimodal approach combining radiotherapy, molecularly targeted agents and surgical resection; however, up to 54% of colorectal cancer patients experience recurrence after combination therapy, and there is an urgent need to develop novel and effective approaches for the treatment of colorectal cancer.
Oncolytic virus (OV) immunotherapy is a novel multifunctional cancer treatment that kills tumour cells while activating systemic immune responses, with oncolytic adenovirus (ADV) being one of the most widely used OVs due to its increasing number of clinical trials as a gene therapy vector. Notably, limited self-replication, host antiviral immunity and the negative immune feedback it triggers have limited the clinical application of ADV. Genetic modification to construct novel therapeutic adenoviruses expressing proteins remains a focus of current research in ADV and is a promising and feasible approach to overcome the therapeutic limitations of ADV.
Neutrophil elastase (NE), a serine protease encoded by the ELANE gene, is normally expressed in primary granules of neutrophils, plays an important role in the coordination of host defence mechanisms and innate immune responses, and possesses unique anti-tumour properties. Relevant studies have shown that uptake of NE by breast cancer cells increases the expression of HLA class I molecules and Cyclin E proteins, and enhances tumour antigen presentation and sensitivity to T cell lysis. In addition, NE and porcine pancreatic elastase (PPE, which is structurally similar to NE) can selectively kill tumour cells and increase effector memory/effector CD8+ T-cell (TEM/TE) infiltration, which can safely and effectively address the problem of insufficient TEM/TE infiltration in immunotherapy.
In this study, the team constructed novel lysogenic viruses expressing NE or PPE, ADVNE and ADVPEE, and demonstrated that they induced colorectal cancer cells to undergo pyroptosis accompanied by the release of HMGB1. HMGB1 binds to TLR4 on the surface of macrophages, activates the MyD88-NFκB-NLRP3 (ASC) pathway, and promotes the M1 polarisation of TAMs, which leads to increased TEM/TE cell infiltration and enhance anti-tumour efficacy. Providing new insights for advancing anti-tumour immunotherapy of lysosomal ADV.
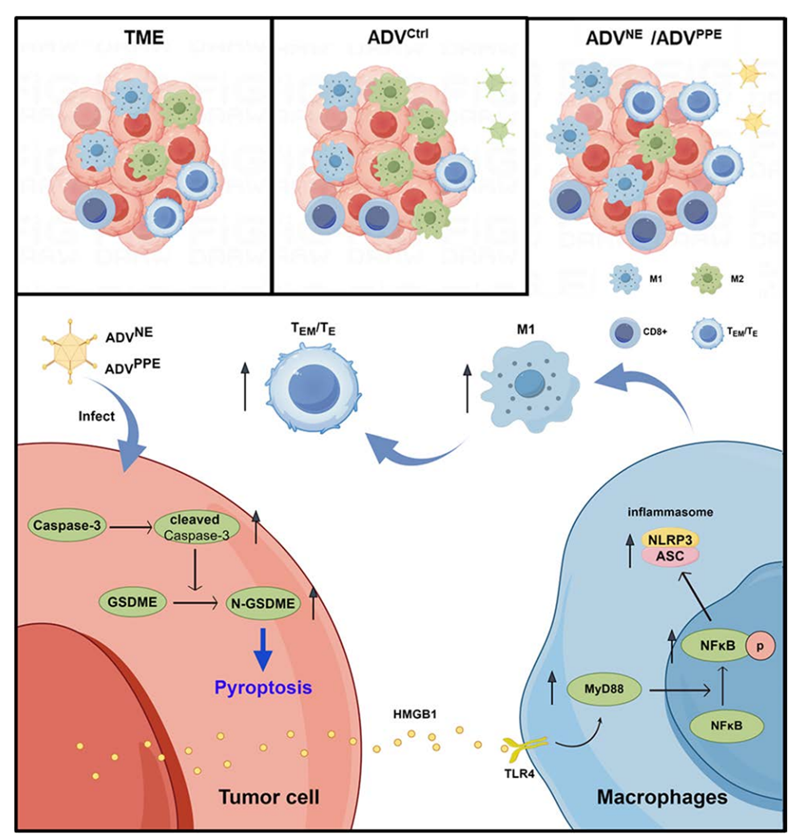
The research team first evaluated the therapeutic efficacy of ADV and changes in the immune microenvironment using subcutaneous tumour models in MC38 and CT26 mice, confirming that ADV has significant anti-tumour properties, but leads to a significant reduction in TEM/TE infiltration and an increase in M2 macrophage infiltration, triggering negative immune feedback.
Data analysis of The Cancer Genome Atlas (TCGA) database revealed that ELANE gene expression was higher in normal tissues than in tumour tissues and that colorectal cancer patients with high ELANE expression had a good prognosis. It has been shown that NE and PPE exhibit strong selective cytotoxicity against a variety of tumour cell lines and significantly increase TEM/TE infiltration in tumour tissues.
Recombinant tumour lysing adenoviruses expressing NE or PPE were subsequently constructed, and in vitro experiments and in vivo experiments in mice confirmed that ADVNE/ADVPEE induced colorectal cancer cell pyroptosis as well as the release of HMGB1 without affecting non-tumour cells, significantly increased tumour lysing activity, prolonged the survival time of mice, and formed memory immunity in the second time that healed mice were inoculated with colorectal cancer cells. With the help of flow cytometry, immune cell depletion and other experiments, it was found that ADVNE or ADVPEE promoted the polarisation of TAM towards the M1 phenotype, which increased the infiltration of CD8+ T cells and TEM/TE cells in the TME and enhanced the anti-tumour efficacy of ADV.
Further exploration revealed that HMGB1-deficient tumour cells were unable to induce M1 polarisation in bone marrow-derived macrophages (BMDMs). Tumour cells treated with ADVNE or ADVPEE released HMGB1 to bind to TLR4 receptors on macrophages, activating the MyD88-NFκB-NLRP3 (ASC) pathway and inducing macrophage M1 polarisation. Finally, the excellent clinical translational potential of ADVNE and ADVPEE was confirmed by a humanised CDX mouse model and preliminary safety assessment results.
Scientific inspiration: This study enhanced the anti-tumour activity of ADV through genetic modification, proposed an effective strategy to improve the therapeutic efficacy of ADV, provided a reliable scientific basis for the development of novel therapeutic lysosomal viruses, and may provide a new perspective for advancing the progress of cancer immunotherapy.
This study was supported by the Shandong Provincial Laboratory Project (No. SYS202202), the National Natural Science Foundation of China (No. 81972888, 82272819), the Provincial Key Research and Development Programme (No. BE2022840), and the Scientific Research Project of Jinan Microecological Biomedicine Shandong Laboratory (No. JNL-2025008B, JNL-2025009B, JNL- 2025011B,JNL-2025010B, JNL2025012B, JNL-2023017D) were funded.
Original link: https://doi.org/10.1186/s13046-025-03358-y